Pier Compliance blog provides in-depth regulatory guides for product safety and compliance in Turkey and the EU: KKDIK, KKS, MBDF, SDS, SEA, EU REACH, GPSR, EPR/PPWR, CBAM, ESPR and Digital Product Passport (DPP), biocidal authorisation and Only Representative strategy. Content is informational, not legal advice.
Featured compliance guides
- What is KKDIK? Turkey REACH guide
- KKDIK interim registration deadline 2026
- KKDIK consortiums, MBDF and LoA
- KKDIK downstream users — 30 September 2026
Regulatory blog — quick answers
- What topics does the Pier Compliance blog cover?
- KKDIK and Turkish chemicals law, EU REACH, SDS, GPSR, EPR/packaging (PPWR), CBAM, ESPR/DPP, biocidal authorisation and market-access guides.
- Where are KKDIK interim registration guides?
- Articles on the 30 September 2026 deadline, KKS data readiness, individual interim registration, pre-MBDF, consortium/LoA and downstream-user duties.
- Is blog content legal advice?
- No. Use articles for horizon scanning and planning; obligations depend on your role, tonnage and portfolio.
- How is the blog different from news?
- The blog offers longer technical depth; news covers short, time-sensitive regulatory updates.
- What should I do after reading a KKDIK article?
- Map your role (manufacturer, importer, downstream user, OR), inventory and tonnage, then contact Pier Compliance via services or contact pages.
Latest blog posts
- KKDIK Downstream Users: What You Need to Know Before 30 September 2026
- KKDIK and Consortiums: Why MBDF, LoA and REACH Data Sharing Matter in Türkiye
- What Is KKDIK? Turkish REACH Registration, KKS, MBDF and Interim Registration Guide
- KKDIK Provisional (Interim) Registration Deadline: Why 30 September 2026 Matters for Companies Supplying Chemicals to Türkiye
- ESPR Digital Product Passport (DPP): A New Data and Compliance Era for Companies Placing Products on the EU Market
- KKDIK Individual Interim Registration 2026: Which KKS Data Must Be Ready Before 30 September?
- How to Calculate CBAM Emissions: A Practical Step-by-Step Guide for Manufacturers
- PPWR and PFAS: How the New EU Packaging Rules Will Impact Companies
- What Is an SDS (Safety Data Sheet) and Who Prepares It in Turkey?
- KKDIK Provisional (Interim) Registration in Turkey: KKS Strategy, Market Continuity and 2026 Timeline
Blog
Latest Articles and News
Expert insights and latest developments on product safety and regulatory compliance.
Explore expert guides on KKDIK, REACH, SDS, GPSR, EPR, CBAM and ESPR/DPP in one place.

KKDIK Downstream Users: What You Need to Know Before 30 September 2026
Before the 30 September 2026 KKDIK interim registration milestone, downstream users should review use notification, SDS alignment, exposure scenarios and supply-chain roles. Pier Compliance supports role mapping and Chemical Assessment Specialist–approved SDS.
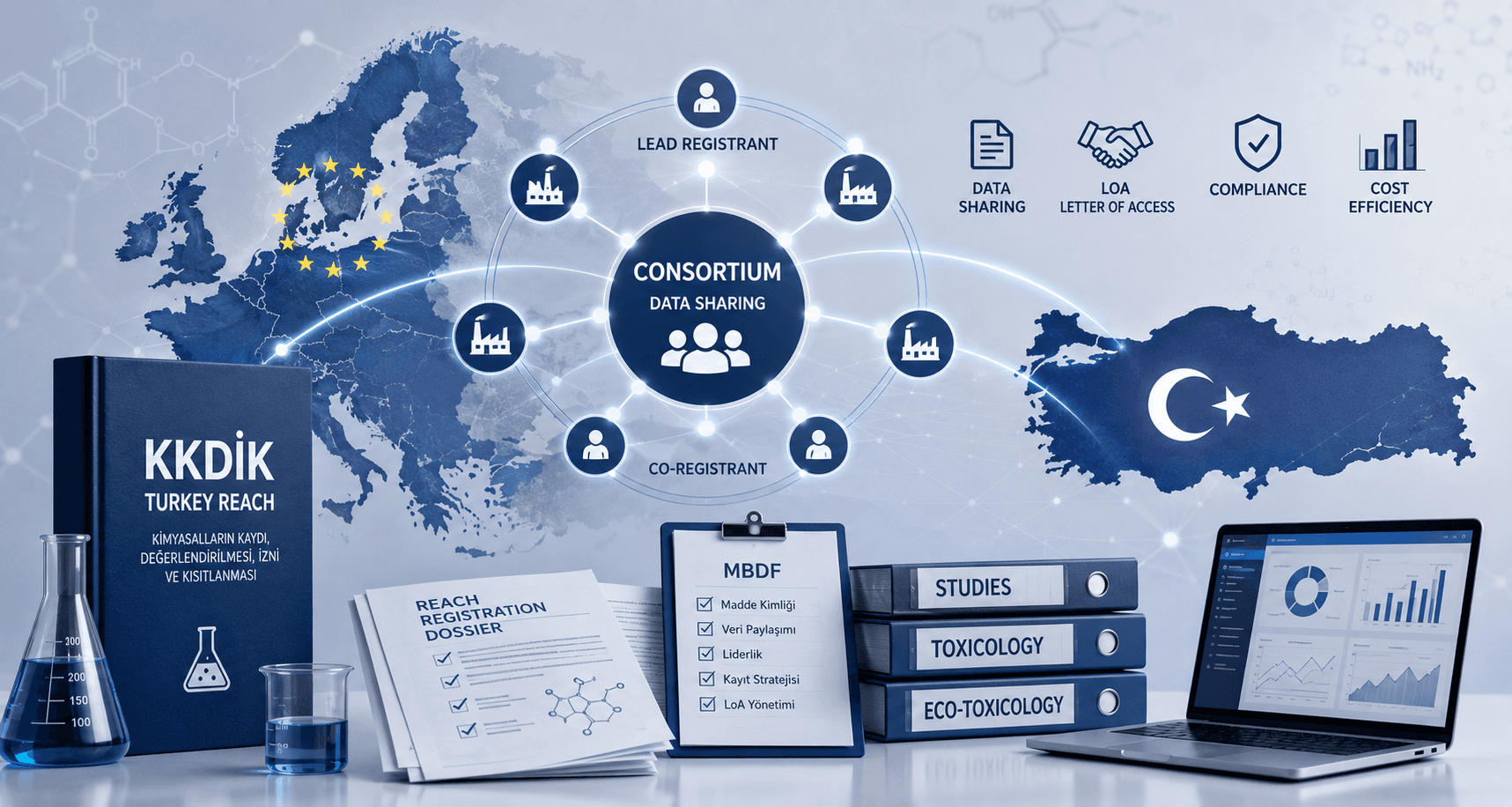
KKDIK and Consortiums: Why MBDF, LoA and REACH Data Sharing Matter in Türkiye
What is a KKDIK or REACH consortium, why is it needed, and how do MBDF, LoA, lead registrant and data sharing affect Turkey REACH registration? Practical guidance from Pier Compliance.
What Is KKDIK? Turkish REACH Registration, KKS, MBDF and Interim Registration Guide
Learn what KKDIK / Turkish REACH means, who must register, how KKS, Pre-MBDF/MBDF, Only Representative, SDS and 30 September 2026 interim registration planning work in Turkey.
KKDIK Provisional (Interim) Registration Deadline: Why 30 September 2026 Matters for Companies Supplying Chemicals to Türkiye
30 September 2026 KKDIK provisional (interim) registration deadline for substances on the Turkish market at ≥1 tonne/year (unless exempt). Editorial Pier Compliance blog visual aligns with KKS, Pre-MBDF, OR and full-registration planning.

ESPR Digital Product Passport (DPP): A New Data and Compliance Era for Companies Placing Products on the EU Market
Under the ESPR, the Digital Product Passport is more than labeling: it reshapes traceability, product data management and EU product compliance. This article summarises how JRC methodological work helps define proportionate Digital Product Passport data requirements.

KKDIK Individual Interim Registration 2026: Which KKS Data Must Be Ready Before 30 September?
A practical guide to KKDIK individual interim registration before 30 September 2026: required KKS data, missing-data justification, and only representative strategy in Turkey.

How to Calculate CBAM Emissions: A Practical Step-by-Step Guide for Manufacturers
How are CBAM embedded emissions calculated? Learn the practical methodology for direct emissions, indirect emissions, precursors, and specific embedded emissions.

PPWR and PFAS: How the New EU Packaging Rules Will Impact Companies
PPWR and PFAS requirements are reshaping EU packaging compliance. We explain the key dates (11 February 2025 and 12 August 2026), EPR links, and practical actions for manufacturers, importers, and exporters.
What Is an SDS (Safety Data Sheet) and Who Prepares It in Turkey?
A corporate guide to Safety Data Sheets for Türkiye: legal role, operational impact, KKDIK and SEA alignment, and why a Chemical Assessment Specialist is central to defensible SDS quality.
KKDIK Provisional (Interim) Registration in Turkey: KKS Strategy, Market Continuity and 2026 Timeline
A practical guide to KKDIK provisional (interim) registration in Turkey: KKS preparation, 30 September 2026 criticality, lead registration scenarios, SEA and pre-MBDF alignment.
5 Critical Risks in KKDIK and SEA Compliance Before 30 September 2026
A practical brief for regulatory teams on KKDIK Turkey and SEA Regulation risks: 1-ton threshold, individual interim registration, polymer/monomer scope, Turkish SDS consistency and technical dossier quality.

Biocidal Product Authorisation in Turkey: Applications, Risk Assessment, and Renewal
A practical walkthrough of biocidal product authorisation—from product type and competent authority to testing, dossier build, e-submission, completeness review, risk assessment, validity, and renewal planning.